Ventilator testing and calibration use lung simulators to verify tidal volume, pressure limits, alarms, and waveform performance under realistic patient conditions. By simulating lung resistance and compliance, biomedical and respiratory teams can confirm ventilators deliver accurate, repeatable performance for safe clinical use.
Lung simulators play a critical role in modern ventilator training for respiratory therapy programs. By allowing educators to adjust compliance and resistance, simulators help students connect ventilator settings to real lung behavior, building confidence, clinical judgment, and hands-on experience before entering patient care.
Respiratory emergencies like ARDS and COPD evolve quickly, demanding rapid clinical judgment and adaptability. Dynamic lung simulation allows educators and care teams to train for changing compliance, resistance, and disease progression—building the confidence and problem-solving skills needed when real patients don’t follow a script.
In medical training, precision matters—but so does endurance. Simulation tools need to perform accurately today, tomorrow, and for years to come. When they don’t, programs stall, lessons lose impact, and confidence fades.That’s why durability is at the heart of every simulator we build at Michigan Instruments.
When Sudden Cardiac Arrest (SCA) strikes, there’s no warning—and no time to waste. Within seconds, the heart stops beating effectively, cutting off blood flow to the brain and vital organs. Survival depends entirely on what happens in those first few minutes. This October, during Sudden Cardiac Arrest Awareness Month…
In healthcare and emergency response, there’s a world of difference between what’s taught in the classroom and what happens in the field. Textbooks and lectures lay the foundation—but real-world emergencies demand fast thinking, precise coordination, and technical confidence…
Cardiac emergencies can strike suddenly and without warning. Whether it’s a heart attack or a cardiac arrest, knowing how to respond in these crucial moments can save a life.
In this blog, we’ll walk you through a step-by-step guide on what to do during a cardiac emergency, emphasizing quick actions and the importance of reliable medical equipment to assist in these life-threatening situations.
Step 1: Assess the Situation
When faced with a potential cardiac emergency, the first step is to assess the situation. Check for signs of cardiac arrest, such as unresponsiveness and abnormal and/or no breathing.
If the person is unresponsive and not breathing, this may indicate a cardiac arrest, which requires immediate action.
Step 2: Call for Help
Dial 911 immediately. This ensures that emergency services are on their way and can provide advanced care as quickly as possible. If you are in a healthcare setting, alert other medical professionals to the situation.
Step 3: Start Chest Compressions
Chest compressions are a critical first step in saving a life during a cardiac arrest. Place the heel of your hand on the center of the chest, just below the sternum.
Use your other hand to support it and begin compressing the chest at least 2 inches deep at a rate of 100-120 compressions per minute. Be sure to allow full chest recoil after each compression.
Step 4: Administer Defibrillation (If Available)
If an automated external defibrillator (AED) is nearby, turn it on immediately and follow the instructions provided. The AED is programmed to analyze the person’s heart rhythm and, if necessary, deliver a shock to restore normal heart activity. Continue CPR until the AED is ready to administer a shock or until emergency personnel arrive.
Step 5: Continue CPR
If an AED is not available, continue CPR until help arrives. If you are trained in advanced techniques, you may also consider performing rescue breaths, especially in cases where the person is a child or an infant.
For untrained responders, hands-only CPR (chest compressions without breaths) is still highly effective in improving survival outcomes.
Step 6: Provide Post-Resuscitation Care
Once emergency responders arrive, they will take over the care and transport the individual to the hospital for further treatment. Follow any additional instructions given by medical professionals, and ensure you remain calm throughout the process.
How Michigan Instruments Helps in Cardiac Emergencies
Cardiac emergencies demand rapid and effective interventions, and the right tools can significantly impact the outcomes. Michigan Instruments has been a leader in life-saving technology since 1964, supporting healthcare professionals and first responders with reliable and easy-to-use solutions.
Our automated CPR devices are designed to deliver precise and consistent compressions, allowing medical teams to focus on what matters most — saving lives. Whether in the field or at the hospital, Michigan Instruments empowers those on the front lines to provide optimal care in critical moments.
With over 60 years of experience, we remain committed to improving patient outcomes and supporting the heroes who respond to cardiac emergencies.
Learn more about how our advanced automated CPR devices can make a difference for your team.

In the early 1960s, a revolutionary concept was born in the unlikeliest of places and changed the world of emergency medicine forever. The groundbreaking idea, nurtured by Clare Barkalow, would later lead to the development of some of the most innovative CPR devices in history and the founding of Michigan Instruments.
The journey began in Grand Rapids, Michigan in the Advanced Engineering Projects department at Lear Siegler, where Barkalow was working as an engineer in 1962. It was here that Barkalow envisioned and developed a device that could automate chest compressions during cardiac arrest.
This invention, known as the External Cardiac Compressor (ECC), was initially tested on dogs and baboons, and the results were astonishing: 24 animals were successfully resuscitated after 90 minutes of fibrillation. These early successes hinted at the potential of automated CPR, but the full vision had yet to be realized.
Going Solo: The Founding of Michigan Instruments
Despite the promising results and groundbreaking potential, Lear Siegler was not interested in pursuing the ECC further. Undeterred, Barkalow took a bold step. In 1963, he left the company and founded Michigan Instruments in Grand Rapids, where he continued to refine his invention, turning his vision into reality. By 1965, the first ECC for human use, the Model 1001, was released, setting the stage for a new era in cardiac resuscitation.
A Perfect Alignment: The Birth of CPR
The late 1960s were a time of rapid development, not only for Michigan Instruments, but for healthcare in general. To put into perspective just how groundbreaking the idea of mechanical CPR was, it should be noted that the ECC was invented at the same time that manual CPR itself was formally endorsed by the American Heart Association (AHA), marking the beginning of a new era in life-saving technology.
The ECC, featuring a pneumatically powered chest compressor, marked a significant advancement with about 100 units being sold across the United States, highlighting a growing recognition of automated resuscitation.
Michigan Instruments seized the momentum. The Model 1001 was soon joined by the Model 1002, a ventilator designed to work alongside the ECC. This paved the way for a unified device capable of both chest compressions and ventilation.
In 1968, the two models were combined to create the Model 1003, initially known as Life-Aid® CPR. Life-Aid® CPR represented a monumental advancement in emergency medical care, as it was the first hands-free CPR system to integrate both chest compression and ventilation. The device was so successful that it gained international attention, being marketed globally under various names, including The Thumper® by Dixie USA and Sier Pulse by Siemens.
Pioneering Advances in Emergency Medicine
Over the next two decades, we continued to refine and improve our devices. For example, the Model 1004, which was introduced in 1974, featured a color-coded system for prescribing chest compression depth based on patient size, further enhancing the precision and effectiveness of CPR.
Clinical studies, like John R. Allen’s “The use of the life aid cardiopulmonary resuscitator– preliminary report” (British Journal of Clinical Practice), began to validate the superiority of mechanical CPR over manual methods. These evaluations of the MII Life-Aid demonstrated clinical safety and the inadequacies of manual CPR, while underlining the advantages of the Life-Aid. These clinical studies helped to solidify Michigan Instruments’ reputation as a pioneer in the field, and soon led to MI listing the device with the FDA.
Clinical Studies Continue to Roll In
- 1978: “Dallas EMS System Advocates Mechanical CPR in Emergency Medical Services” was published in July and August of 1978 by Bill G. Roberts and Jane M. Bryan. It showed how favorable the results of using the mechanical CPR devices were over manual CPR.
- 1978: “External cardiac compression. A randomized comparison of mechanical and manual techniques” was published by Taylor, George J.; Richard Rubin; Michael Tucker; H. Leon Greene; Michael T. Rudikoff; and Myron L. Weisfeldt, and found that MI mechanical CPR compares with best of manual CPR and is indicated for longer term resuscitation or when manual CPR is difficult to perform.
- 1979: “Machine vs. manual cardiopulmonary resuscitation in moving vehicles” (Roberts, B.G.) was published in The EMT Journal, showing that MI mechanical CPR performs better CPR in a vehicle than manual CPR. It also showed fewer interruptions, plus a constant depth of compression with no pause, no risk of fatigue, with less personnel, and the ability to be more patient-accessible.
- 1979: “Clinical assessment of patients undergoing CPR in the emergency department”, published in JACEP by Lilja, G. Patrick; Martin Hill; Ernest Ruiz; and Joseph Clinton found that MI mechanical CPR performs well over long resuscitations and calms down hectic cardiac arrest situations.
- 1982: “Mechanical cardiopulmonary resuscitation” (Harmon, Annette L.) is published in Medical Instrumentation. At this point, MI mechanical CPR has been in use for 20 years and is proven more effective with less patient injury.
These clinical studies demonstrated the superiority of mechanical CPR over manual CPR, especially during prolonged resuscitation efforts or in challenging environments like moving vehicles. They also aided to further prove that it is a valuable tool in defining effective CPR, including AHA Guideline protocols.
The Innovations Continue
The 1970’s and 1980s saw Michigan Instruments expand product lines and continue to push the boundaries of what was possible in CPR technology. These innovations further demonstrated our commitment to advancing both training and practical application in the medical field.
- 1976: Michigan Instruments recognized the need for comprehensive training and simulation in respiratory care, and released the Dual Adult Training & Test Lung under the trademark TTL® and VentAid®
- 1982: The Adult Infant Lung was developed and released under the trademark LifeSpan®
- 1984: The Programmable Thumper®, based on a 6502 embedded microprocessor system, was developed. It incorporated a microprocessor system and set a new standard for CPR research tools, helping to define effective CPR protocols.
- 1985: Model 1005 was released with an improved pneumatic control system, increased capacity for patient size, and increased adjustable compression force.
These advancements not only improved patient outcomes but also highlighted the limitations of manual CPR—such as rescuer fatigue and inconsistent compression depth—solidifying the importance of mechanical solutions in emergency care.
Expanding Horizons for Global Impact
We didn’t stop there; Michigan Instruments’ first trip to Japan in 1988 kicked off a field study and the introduction of the Thumper® to the medical community, expanding our reach to a global market. Over the next couple of decades, we continued to innovate, introducing new models and expanding our reach globally.
- 1991: PneuView® DOS software with electronic instrumentation was integrated into the TTL product line.
- 1997: PneuView® Windows software was introduced. It was also in this year that the single lung TTL was released.
- 2000s: MII’s products were in use not only in the United States but also in countries like Japan, France, and Ireland.
- 2005 – Introduction of the Thumper 1007CCV and Thumper 1007CC
- 2008: The introduction of the Life-Stat® Model 1008, featuring an electronic control system, marked another leap forward in automated CPR technology.
- 2014: The PneuView and TTL products were updated. Updating the design and functionality of the device and particularly the Pneuview software.
- 2022: The Spontaneous Breathing Lung was introduced
With innovations and software integrations like PneuView®, our lung simulation products became essential tools for teaching, training, and equipment testing in universities, hospitals, and EMS services worldwide.
A Legacy of Life-Saving Technology, Innovation and Excellence
As a company, Michigan Instruments stands as a testament to the power of innovation and perseverance. From Clare Barkalow’s initial idea in 1962, our devices have been instrumental in advancing CPR practices, aligning perfectly with the evolution of CPR guidelines over the decades.
Today, Michigan Instruments continues to impact global markets, including the USA, China, Europe, India, Brazil, and the Middle East. Our product lines have expanded to meet the diverse needs of healthcare professionals in many areas, emphasizing both quality and innovation.
Join the Revolution in CPR Practices
For over half a century, Michigan Instruments has been revolutionizing the way the world approaches cardiac resuscitation. Our automated CPR devices have proven time and time again to offer superior performance, reliability, and outcomes compared to manual methods.
Isn’t it Time You Upgraded your CPR Practice?
For healthcare professionals looking to revolutionize their own CPR practices, we offer decades of proven expertise and cutting-edge technology. To this day, we are inspired by Barkalow’s determination to bring his idea to life, despite his employer declining to pursue it, and will continue to be at the forefront of developing lifesaving technology.
With Michigan Instruments on your team, you can ensure that you’re equipped with the best tools for saving lives.
Partner with us to enhance your emergency response capabilities, improve patient survival rates, and stay ahead with cutting-edge technology that’s been trusted for decades.
Contact Michigan Instruments today to learn how our automated CPR devices and lung simulation products can transform your approach to emergency care.

Technological advancements continue to shape the way professionals acquire critical skills in medical education and research.
One such innovation that stands out is the integration of respiratory training simulators, specifically lung simulators, into the educational toolkit of medical professionals, ventilator manufacturers, and educators.
Among the pioneers in this field is Michigan Instruments, offering state-of-the-art lung simulators paired with PneuView Software.
What Sets Our Lung Simulators and PneuView Software Apart?
Michigan Instruments takes pride in providing lung simulators that are fully to scale, offering realistic residual lung volumes and capacities. The range of settings for compliance and resistance on TTL® and PneuView® systems surpasses most other available simulators. What makes these devices truly exceptional is their ability to move and “feel” like real lungs when ventilated.
Unlike basic test lungs that perform only a handful of simulations and lack full scalability, Michigan Instruments’ lung simulators offer a superior level of realism.
This realism is not just a feature; it is a crucial aspect that enhances the utility of these devices for hands-on training, product testing, and research and development.
Unparalleled Realism for Lifesaving Education
The combination of Michigan Instruments’ Lung Simulators and the PneuView Software creates a more realistic environment for medical professionals and students.
These devices go beyond basic simulations, providing an experience that more closely replicates the complexities of the human pulmonary system.
The realistic representation of adult and infant lung conditions allows for a diverse range of simulations, enhancing understanding and clinical skills development.
Why Choose Michigan Instruments?
There are several reasons to choose Michigan Instruments, just as facilities around the world have.
Pioneer in Respiratory Care and Emergency Medical Industries
Michigan Instruments has established itself as a pioneer in the respiratory care and emergency medical industries.
Our lung simulators and automated CPR devices have been recognized as breakthrough innovations in the medical profession.
Gold Standard of Respiratory Simulation
With thousands of users worldwide, Michigan Instruments’ lung simulators are considered the gold standard of respiratory simulation.
The devices have proven their efficacy in various applications, from medical education to testing to research and development.
Applications Across Industries
Classroom Simulation: Michigan Instruments’ TTL and PneuView Lung Simulators facilitate “Aha Moments!” in classrooms, offering hands-on experiences that enhance understanding and develop valuable clinical skills in students.
Research and Development: The lung simulators play a crucial role in the design, engineering, evaluation, and manufacturing of respiratory devices. They contribute to making these devices more user-friendly and effective.
Testing: Evaluate the performance of respiratory devices, even in the face of changing pulmonary dynamics. PneuView Systems enable the collection and display of ventilation data through advanced software.
Request a Quote: Join the Ranks of Professionals Choosing Excellence
Discover why thousands of educators, researchers, manufacturers, and quality assurance professionals worldwide trust Michigan Instruments’ lung simulation devices.
Experience high-quality lung simulators that you can touch, see, and modify to meet the evolving demands of respiratory care and medical education.
Michigan Instruments is at the forefront of transforming respiratory training in the digital age, providing cutting-edge solutions that bridge the gap between theory and practice in the lifesaving field of respiratory care.
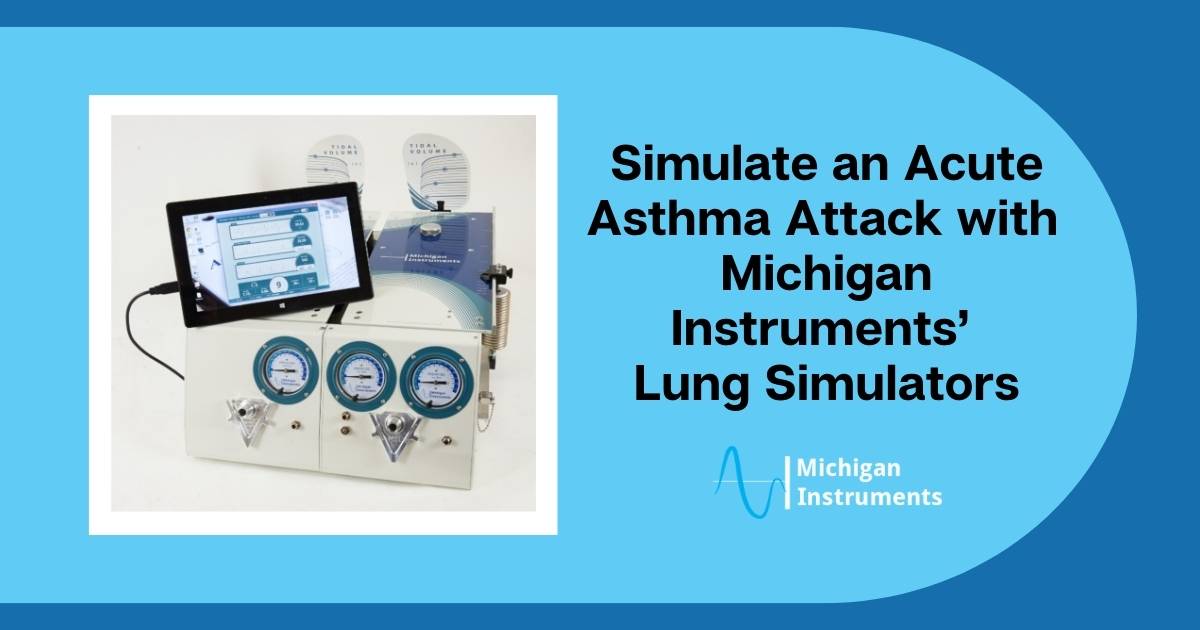
Asthma is a chronic condition that inflames and narrows the airways in the lungs. In fact, nearly 26 million people in the U.S. have asthma, making it one of the most common forms of respiratory disease in the country.
Fortunately, you can easily replicate asthma attack simulation on a lung simulator for education and research purposes.
Why Use Michigan Instruments Lung Simulation for Learning about Asthma?
There are several reasons why our Lung Simulators are a fantastic tool for learning and teaching about asthma; including, but not limited to:
- Safety: Simulation-based education ensures patient safety. This allows the user to make mistakes and gives them the ability to correct them, without harm to a patient.
- Reduced Errors: Simulation provides opportunity for continued practice. This results in less error while working with humans.
- Preparation for Several Scenarios: Simulation devices offer a wide range of lung compliance and airway resistance settings. This offers the students and professionals an immersive and nearly hands-on experience.
Settings For An Asthma Attack Simulation
By creating a severely restricted airway and pairing it with a slightly compliant lung, you can provide settings that simulate an acute asthma attack.
Here are the settings we recommend (using our lungs) to create a successful simulation:
- Functional Residual Capacity: Should be between 860mL and 990mL per adult lung and 80mL to 200mL for the infant lung.
- Please note that residual capacity is only considered functional if it’s downstream (during inspiration) of the airway resistor.
- Airway Resistance: A resistance of 50cmH2O/L/s should be used to simulate this condition in adults. The user should use 500cmH2O/L/s to simulate this condition in infants.
- Dynamic Compliance: Use a lung compliance of .04L/cmH2O for adults and .004L/cmH2O for infants. It’s important that this value is “dynamic” so that the compliance of the simulated lung will decrease as breaths begin to stack.
How is This Simulation Beneficial?
During normal ventilation, the increased airway resistance creates the high proximal pressure seen in victims of an acute asthma attack.
Additionally, the above-average compliance of the simulated patient will cause breath stacking unless the breaths delivered are properly regulated.
You can test many simulations in a number of ways. This allows students to get hands-on experience with rare conditions, replicate case-specific failures in ventilators, identify pros and cons of different ventilation techniques and more.
Organizations around the world use our test lungs for a plethora of educational purposes. The flexibility of our equipment allows our devices to replicate hundreds of healthy and diseased lung conditions, like asthma, while providing accurate measurements and data that inform your testing, research, and training.
Learn More About Our Lung Simulation Capabilities
If there is a specific symptom or condition that you would like to replicate, please let us know. We’d love to put together a simulation for you.
For questions or more information about simulations, contact us today.

